The following module was designed to supplement medical students’ learning in the clinic. Please take the time to read through each module by clicking the headings below.
By the end of the tutorial, the following objectives should be addressed:
Malignant Spinal Cord Compression (SCC) is an oncologic emergency defined as the radiographic compression of the spinal cord or cauda equina that occurs as a result of metastatic or spinal tumor growth that either directly or indirectly causes impingement [1 - 3]. The most common presenting symptom is pain, with severity of symptoms varying depending on the degree of compression from asymptomatic to frank paraplegia, which may be reversible or irreversible [3]. Spinal cord compression can arise from primary spinal tumors or any metastatic tumor with a tendency to spread to the vertebral column [2]. Complications include pain and potentially irreversible neurologic dysfunction that may severely impact patients’ functional ability and overall quality of life [1].
There have been no recent studies investigating the incidence of SCC due to variation in definitions, and few studies report trends in incidence rates. A systematic review found that 2.8% of patients with spinal metastases eventually develop spinal cord compression (SCC) [4]. Consequently, as the incidence of solid tumours increases and survival rates improve, more patients are at risk of both spinal metastases and SCC. In adults, the majority of SCC cases arise from metastases of lung, breast, and prostate cancer. On the other hand, SCC is observed in 5% of pediatric cancer patients and is most commonly caused by Ewing sarcoma and neuroblastoma [3]. Most recent results from a Canadian population-based study reviewed incidence, management, and outcome of SCC, and demonstrated an overall 2.5% cumulative probability of experiencing at least one episode of SCC in the five years preceding death from cancer. Cumulative probability ranged from 0.2 - 7.9% depending on the primary tumor site, with highest cumulative incidence seen in myeloma (7.9%), prostate (7.2%), nasopharynx (6.5%), and breast malignancies (5.5%). Other studies suggest an incidence of SCC up to 6% based on autopsies and other reports [2].
Surrounded by the dural thecal sac, the spinal cord extends from the foramen magnum to L1-L2 in adults. In children, the spinal cord extends more inferiorly (L2-4) [3]. The spinal cord is divided into four longitudinal regions (cervical, thoracic, lumbar, and sacral) each comprising multiple spinal cord segments. Exiting from the intervertebral foramina, each segment has two pairs of spinal nerve roots which mediate motor and sensory functions [5]. The cauda equina is defined as the lumbar and sacral spinal nerves located in the lumbar cistern from L1-L2 to S2.
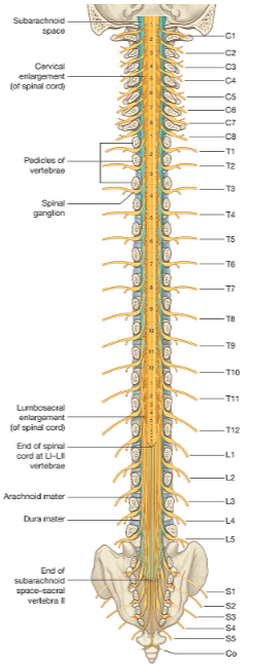
SCC occurs primarily through two main mechanisms - (1) external compression by a tumour arising from the vertebral body and (2) internal compression due to intramedullary metastasis [3]. Tumor metastases to the bony elements of the vertebral column account for 85-90% of SCC [2].
Possible mechanisms of metastases include [2]:
Expansion of the tumor within the spinal column compromises epidural venous plexus flow, leading to vasogenic edema of white and gray matter; if uncorrected, infarction eventually ensues [2].
80-90% of SCCs are metastatic tumors localized in vertebral bodies, causing compression of the anterior/anterolateral aspects of the spinal cord, often yielding localized symptoms [1, 2].
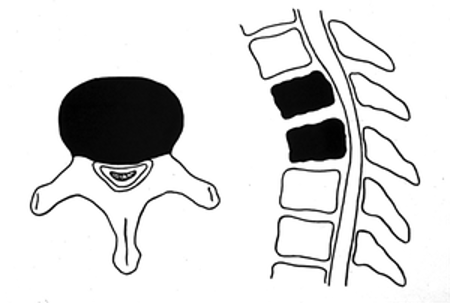
10% of SCCs are paraspinal masses that invade through the neural foramen and into the epidural space [1, 2].
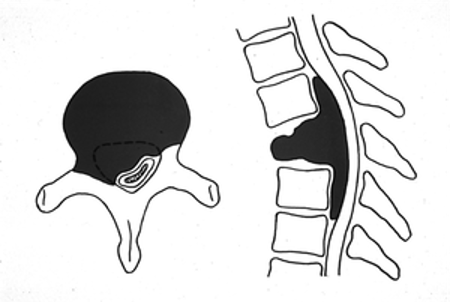
The thoracic spine is most commonly affected due to its larger vertebral surface area and greater propensity for metastatic disease, with 60% of SCC localized to the thoracic spine, 30% to the lumbosacral spine, and 10% to the cervical spine [1, 2].
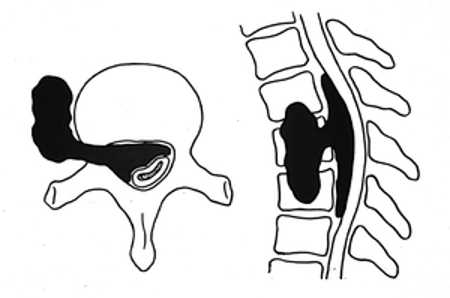
Spinal cord compression (SCC) can be further classified as intramedullary, leptomeningeal, or extradural, depending on the tumor site [1]. Malignant extradural SCC occurs when the tumor invades the epidural space, compressing the thecal sac [1].
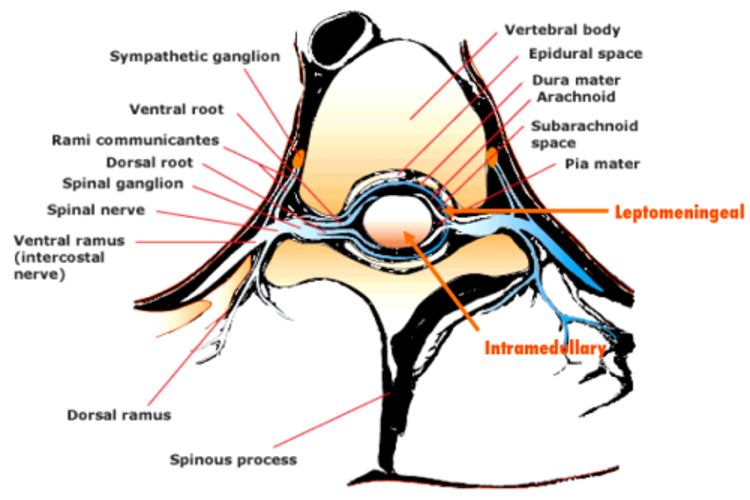
Specific signs and symptoms of SCC vary depending on the level and extent of cord obstruction.
Back pain is the most common presenting symptom in 83-95% of patients, typically most pronounced at night or early in the morning when adrenal steroid secretion is at its lowest [3]. Back pain often precedes neurologic symptoms by weeks, often leading to delayed recognition and diagnosis of SCC [2]. Additional features of the back pain may provide clues to the underlying pathophysiological mechanisms. For example [2]:
60-85% of patients present with weakness that typically worsens, progressing to gait dysfunction and paralysis by the time of diagnosis [2]. The presentation of motor symptoms varies depending on the location of cord compression [1,2]:
While less common than motor findings, sensory changes are nonetheless present in ~50% of patients. Common sensory symptoms include ascending, “band-like”, or “saddle” numbness/paresthesias [2].
Disruption of bladder and bowel function is a late manifestation of SCC observed in up to 50% of patients, with urinary retention being the most common, but rarely the only autonomic symptom [2].
Back pain with neurologic findings has a wide array of etiologies and requires a thorough investigation, beginning with a comprehensive history. History should include general health, past medical history, prior back issues/surgery, characteristics and duration of back pain, exacerbating factors, associated motor/sensory symptoms, and autonomic dysfunction (e.g., bowel/bladder symptoms). A full physical exam should be performed, including a thorough, focused neurologic/MSK exam and an examination of the area of prior malignancy (e.g., breast, lung, abdomen).
Diagnosis of SCC requires radiologic evidence of extrinsic neoplastic compression of the dural sac at the level of the clinical features [1,2]. Thorough imaging of the entire spinal cord, dural sac, and epidural space is critical for diagnosis and detection of additional sites of disease, which significantly impacts management planning and prognostic determination [1,2]. Modalities used for radiologic confirmation include [2]:
Indicated for patients who are not surgical candidates and have an undiagnosed primary cancer, new oligometastasis, or if there is a discordance between the primary lesion and spinal lesion; however, if other sites are identified that would be more amenable to biopsy, then more preferable as bone biopsies do not have a great diagnostic yield [3].
Rapid diagnosis and treatment of SCC is critical in preventing progression of neurological sequelae [1,7]. The goals of treatment should be to improve or maintain the highest quality of life possible (e.g., pain relief, prevention of complications, and restoration/maintenance of the level of neurologic function) [1]. Decisions regarding treatment should consider medical status, ambulatory status, structural factors, anticipated outcomes, treatment goals, and patient preferences [1]. Management of SCC begins with immediate glucocorticoid administration in almost all patients, as glucocorticoids, unlike other modalities, are first-line due to their rapid effect in relieving swelling of the spinal cord. Additional therapies include concurrent symptomatic treatment, followed by definitive treatment (either surgery, radiation therapy, or systemic therapy) [1,7].
Early initiation of high-dose corticosteroids is standard management of SCC. Typically, patients are started on a loading dose of 10mg dexamethasone, followed by 4mg q6 hours, with gastric protection from H2 blockers or proton pump inhibitors such as pantoprazole 40mg daily [8]. Several studies have evaluated the benefit of steroid dose escalation with ultra-high doses (96-100mg vs 10-16mg of dexamethasone) and have demonstrated no benefit with respect to pain control, ambulation rates, or neurologic outcomes [3]. Higher doses are associated with significant toxicity (GI bleed, psychosis, hyperglycemia) without a proven benefit in functional recovery [9]. The duration of the steroid taper should be determined based on the severity of symptoms, clinical response, and definitive management [3]. The ability of corticosteroids to transiently improve neurologic function in patients with SCC, presumably via anti-edema effects and/or oncolytic effects in steroid-responsive malignancies, has long been recognized [7]. When prescribing high-dose corticosteroids, it is important to individualize these therapies and consider patients’ comorbidities such as active infection, uncontrolled diabetes, high-risk gastrointestinal bleeding, etc. Other important therapies to consider are opioid analgesics for further back pain management, venous thromboembolism prophylaxis (unless immediate surgery is anticipated), and catheterization for urinary retention [7].
Selecting the modality of definitive treatment requires the consideration of factors such as [7]:
Depending on the patient’s clinical situation, definitive treatment can include surgery, radiation therapy, and/or systemic therapy. Table 1 outlines several factors to consider when deciding between surgery and radiation therapy.

An important component of the decision-making process when considering definitive therapy is assessment of spinal stability, which refers to the integrity of the vertebral spine to resist progressive deformity and/or neurologic compromise under physiologic loads [2]. Stability is best assessed by a spine surgeon based on radiographic features and clinical symptoms. The Spine Instability Neoplastic Score (SINS) takes into account six different factors of clinical and radiographic findings with a score of >7 warranting surgical consultation [3].

The Neurologic, Oncologic, Mechanical andSystemic (NOMS) framework is a structured decision-making model used to guidethe management of spinal cord compression.
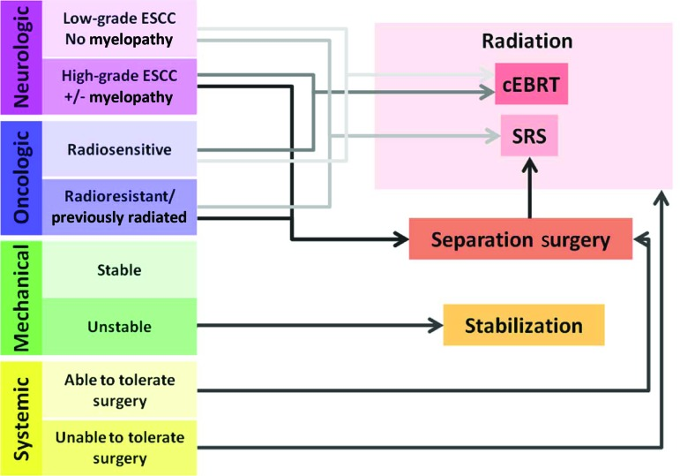
Surgical treatment of SCC is considered in patients with spinal instability and radioresistant tumors that compress on the cord [7]. Treatment entails aggressive tumor resection, and if necessary, followed by spinal reconstruction/stabilization. Surgery provides immediate relief of compression and is beneficial when a histologic diagnosis is unknown, in a previously irradiated site of compression, or when a patient has progressive neurologic deterioration with poor response to steroids [3].
Radiation therapy is a well-established modality for the treatment of SCC and is particularly effective in reducing pain, restoring functional status, and improving sphincter function [1]. If surgical decompression of the SCC is necessary, patients still need post-operative RT to address the underlying cause (i.e., cancer) by providing local control, preventing further progression, and stabilizing surgical instruments during healing. Two RT techniques are used to treat SCC: External Beam Radiation Therapy (EBRT) and Stereotactic Body Radiation Therapy (SBRT).
EBRT is indicated for patients who are considered unsuitable for surgery with relatively radiosensitive tumors (e.g., breast, prostate, ovarian, SCLC, myeloma, lymphoma), and as subsequent treatment following surgical decompression. The optimal dose and schedule of EBRT is debated, and treatment decisions should be tailored to each patient but it typically involves 5 fractions of treatment. Several small randomized trials conducted primarily in patients with solid tumors and a relatively short estimated life expectancy (i.e., 3-6 months or less) indicate that shorter courses of EBRT offer similar palliation compared to longer courses [7]. Response to EBRT is generally excellent for local tumor control and palliation of pain. While typically well tolerated, irradiation to extensive spinal segments may lead to complications such as bone marrow suppression and gastrointestinal toxicity [7].
SBRT, also referred to as Stereotactic Radiosurgery, is a technique that delivers precise radiation to the tumor while reducing irradiation of the surrounding tissue. The targeted nature of SBRT allows the utilization of high-dose radiation that would otherwise be intolerable to the spinal cord, which is a major limitation of EBRT. A further advantage of SBRT is its efficacy in reducing pain and achieving local tumor control in relatively radioresistant neoplasms (e.g., renal cell carcinoma, melanoma, and sarcoma), making the technique an excellent choice for patients with radioresistant cancer [7].
Although most malignancies that cause SCC are chemoresistant, chemotherapy is a potential treatment option in patients with chemosensitive neoplasms. Systemic therapy may require several days or weeks to take effect, and patients with SCC generally require more expeditious tumor treatment, necessitating local therapy such as radiation and/or surgery [7]. Tumors in which chemotherapy has been used in selected cases to successfully treat SCC include Hodgkin lymphoma, non-Hodgkin lymphoma, neuroblastoma, germ cell neoplasms, and breast cancer [7].
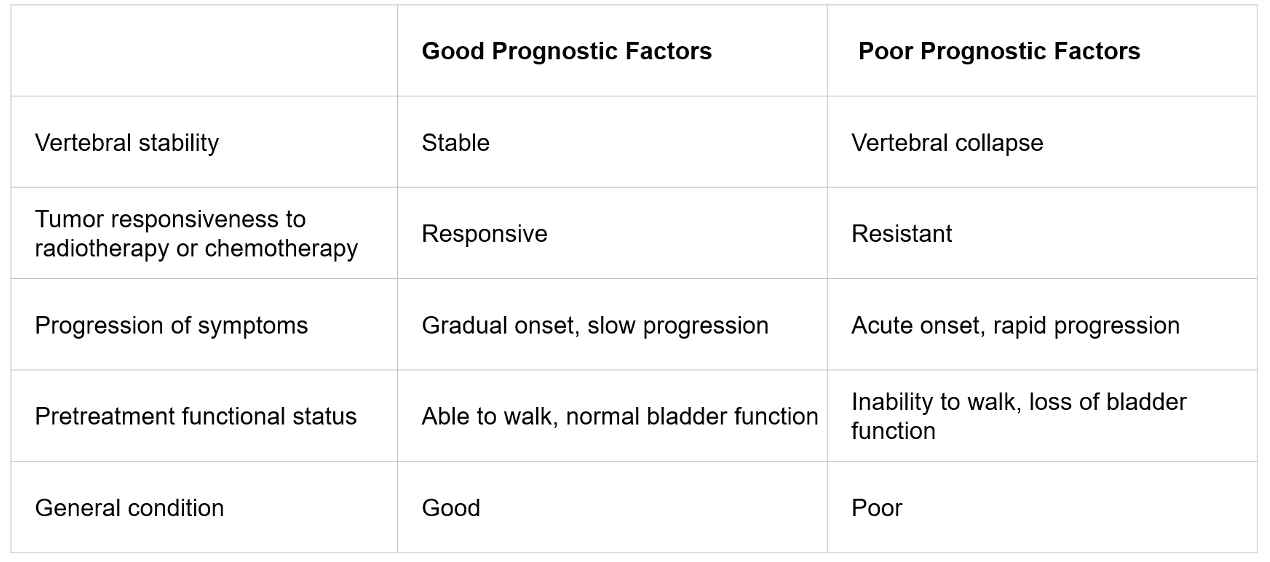
The median survival following diagnosis of SCC ranges from approximately 3 to 6 months [4, 7, 12]. Population-based data from Ontario report a median of 2.9 months after the first episode of SCC [11]. The single most critical prognostic factor for the recovery of ambulation after treatment of SCC is the patient’s neurologic status prior to initiating management [10]. If patients experience a loss of neurological function more than 24-48 hours after onset, the chances of recovery are low, and their initial level of function may become their new baseline. Rehabilitation through physiotherapy and occupational therapy is essential for maintaining or enhancing neurological function. Additional prognostic factors for the outcome of spinal cord compression are outlined in Table 2.
Malignant spinal cord compression is an oncologic emergency and a common sequelae of cancer. Complications include pain and potentially irreversible neurologic dysfunction. The most common presenting symptom is back pain, followed by neurological symptoms, such as motor weakness and sensory changes. The most important steps in the management of SCC rely on expedient diagnosis and treatment of SCC, which depends on the interdisciplinary cooperation of multiple specialties, including:
Initial therapy for SCC should include the immediate administration of glucocorticoids alongside pain and symptom management. Definitive treatment options for SCC include surgical debulking, radiation therapy (EBRT or SBRT), and chemotherapy. Treatment decisions should be individualized based on patients’ functional status, medical comorbidities, structural factors, anticipated outcomes, treatment goals, and patient preferences.
The median survival following diagnosis of SCC ranges from approximately 3 to 6 months [4, 7, 12]. Population-based data from Ontario report a median of 2.9 months after the first episode of SCC [11]. The single most critical prognostic factor for the recovery of ambulation after treatment of SCC is the patient’s neurologic status prior to initiating management [10]. If patients experience a loss of neurological function more than 24-48 hours after onset, the chances of recovery are low, and their initial level of function may become their new baseline. Rehabilitation through physiotherapy and occupational therapy is essential for maintaining or enhancing neurological function. Additional prognostic factors for the outcome of spinal cord compression are outlined in Table 2.
Malignant spinal cord compression is an oncologic emergency and a common sequelae of cancer. Complications include pain and potentially irreversible neurologic dysfunction. The most common presenting symptom is back pain, followed by neurological symptoms, such as motor weakness and sensory changes. The most important steps in the management of SCC rely on expedient diagnosis and treatment of SCC, which depends on the interdisciplinary cooperation of multiple specialties, including:
Initial therapy for SCC should include the immediate administration of glucocorticoids alongside pain and symptom management. Definitive treatment options for SCC include surgical debulking, radiation therapy (EBRT or SBRT), and chemotherapy. Treatment decisions should be individualized based on patients’ functional status, medical comorbidities, structural factors, anticipated outcomes, treatment goals, and patient preferences.
Use your mouse to click through the slides and answer each question in the text box provided.
Note: This case can be completed on an iPad. To do this download the (free) Articulate Mobile Player for the iPad by clicking here.
