The following module was designed to supplement medical students’ learning in the clinic. Content was developed in accordance with the ‘Oncology Goals and Objectives for Medical Students’ established by the Canadian Oncology Education Group (1).
Upon completion of this module, students should be able to:
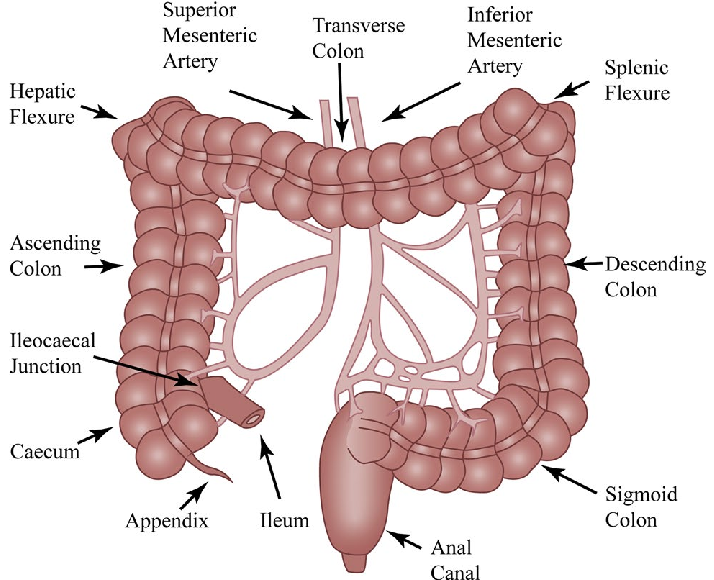
The large intestine consists of the portion of the gastrointestinal tract distal to the ileocecal canal. It consists of the cecum, colon, rectum, and anal canal (1). The colon is further divided into segments: the ascending colon, transverse colon, descending colon and sigmoid colon (1).
The large intestine functions to absorb water and nutrients, and to eliminate waste from the body in the form of stool.
The colon and rectum attached to the wall of the abdominal cavity by a mesentery composed of fatty connective tissue. This mesentery contains the blood vessels, nerves, lymph nodes and lymph vessels serving the large intestine.
Moving from the lumen of the bowel outwards, the bowel wall consists of the following layers (1):
The innermost lining of the bowel wall. It is composed of epithelial cells, a thin layer of connective tissue called the lamina propria, and a thin layer of muscle called the muscularis mucosa.
A layer of connective tissue that lies deep to the mucosa. It contains mucous glands, blood vessels, lymph vessels and nerves.
A thick layer of muscle that lies deep to the submucosa. It is composed of an inner ring of circular muscle fibres and an outer ring of longitudinally-arranged muscle fibres. It is responsible for the peristaltic action of the large intestine.
In the colon, the outer layer of the muscularis propria is arranged into three longitudinal bundles of muscle called the taenia coli. These muscle bundles contract lengthwise to form sac-like pouches along the length of the colon; these pouches are called haustra.
Surrounds the muscularis propria, and is composed largely of adipose connective tissue. It is also referred to as ‘pericolic fat’.
The outermost layer of the colon and is composed of connective tissue.

The rectum is approximately 12 cm in length (2). The upper third of the rectum is covered by peritoneum on the anterior and lateral aspects. The middle third is covered by peritoneum on the anterior aspect only. The peritoneum is reflected laterally to form the perirectal fossa, and anteriorly to form the rectovesicular fold (in men) or rectouterine fold (in women). The lower third is not covered by peritoneum at all, and is referred to as the rectal ampulla (2).
The true anatomical boundary between the colon and rectum is the point at which the taenia coli fuse to the circumferential longitudinal muscle of the rectum. However, for the purpose of determining treatment options, the division between the colon and rectum is the point of peritoneal reflection.

Colorectal cancer is the third most common malignancy in Canada for both men and women (1). The incidence and mortality of colorectal cancer is low until middle-age, but rises rapidly thereafter (2). Colorectal cancer is more common in men than in women, with a lifetime probability of developing colorectal cancer of 7.5% for men and 6.4% for women (1).Colorectal cancer incidence is increasing in young people for unclear reasons, while its incidence in those over the age of 50 remains stable.(3)
Colorectal cancer is also the second leading cause of cancer-related death, exceeded only by lung cancer (1), with 12% of all cancer-related deaths caused by colorectal cancer (4). In Canada, 1 in 29 men and 1 in 34 women are expected to die from colorectal cancer (4).
Age is the most important risk factor for developing colorectal cancer, with risk being significantly higher in patients over 50 years of age (1). The majority of colorectal cancers occur in patients aged 50 years or older who are otherwise of average risk and have no significant family history (1).
The majority of cases of colorectal cancer are sporadic. Only 15-30% of patients with colorectal cancer appear to have a major hereditary component (4). Nevertheless, the risk of developing colorectal cancer is significantly higher in individuals with a family history of colorectal cancer or a known genetic syndrome associated with colorectal cancer.
Individuals with a single first-degree relative who was diagnosed with colorectal cancer before the age of 60 years have a significantly elevated risk of developing colorectal cancer themselves (1,4). The risk is similarly increased for individuals who have two or more first-degree relatives diagnosed with colorectal cancer at any age (1). The risk is not significantly increased in individuals with a single affected second-degree relative (1). Family history affects screening protocols, and it is important to ask patients about their relatives’ history of colon cancer.
Hereditary colorectal syndromes are associated with about 5% of cases of colorectal cancer. The most prevalent predisposing genetic syndromes for colorectal cancer are familial adenomatous polyposis (FAP), and hereditary non-polyposis colorectal cancer (HNPCC). However, additional uncommon genetic syndromes have also been associated with an increased risk of colorectal cancer, including Peutz-Jeghers syndrome and juvenile polyposis syndrome (4).
FAP accounts for 1% of all colorectal cancers (4,5). FAP is inherited in an autosomal dominant pattern, and is caused by a mutation in the adenomatous polyposis coli (APC) gene (4). APC is a tumour-suppressor gene; a loss-of-function mutation in this gene therefore results in a decreased ability of the body to prevent the development of tumours. As a result, individuals with FAP develop hundreds to thousands of adenomatous polyps in their colon. Although these polyps are initially benign, the majority of individuals with FAP will develop colorectal cancer by the age of 30 if a prophylactic colectomy is not performed (4).
FAP is thought to result in colorectal cancer via a ‘two hit model’. The inherited mutation in APC represents the first ‘hit’, and the second hit is usually brought about by a sporadic mutation that occurs in the remaining ‘normal’ allele of the APC gene (5). Further sporadic mutations occurring in these APC-mutated cells are much more likely to lead to cancer than they would be in normal non-mutated cells.
HNPCC, also known as Lynch syndrome, is the most common hereditary colorectal syndrome, and accounts for 3% of all colorectal cancers (5). It is inherited in an autosomal dominant pattern, with an 80% penetrance, and is caused by a mutation in a DNA mismatch repair gene. Like FAP, HNPCC follows the two hit model, and thus requires the combination of an inherited mutation and a secondary sporadic mutation to result in malignancy. The presence of loss-of-function mutations in both alleles of the mismatch repair gene results in microsatellite instability; this is a term referring to the contraction and expansion of areas with repetitive nucleotide sequences (microsatellites) (5).
Unlike FAP, which is characterized by the growth of thousands of polyps, patients with HNPCC may have only a normal or mildly increased number of polyps (4). However, patients with HNPCC tend to develop polyps at an earlier age, and the polyps they develop are more likely to become cancerous (4). The mean age at diagnosis of colorectal cancer in patients with HNPCC is 43 years (5).
HNPCC is divided into two subtypes. HNPCC type I is characterized by an increased risk of colorectal cancer only, whereas HNPCC type II is associated with an increased risk of colorectal, endometrial, ovarian, gastric, small bowel, bile duct, urinary tract, and skin malignancies (4,5).
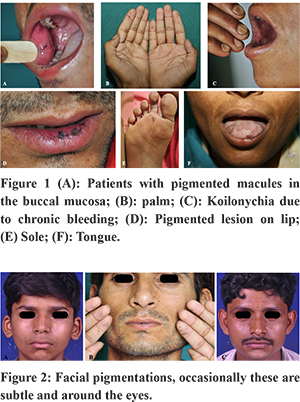
Peutz-Jeghers syndrome is associated with less than 1% of colorectal cancers (5). It is a rare autosomal dominant disease with high penetrance, characterized by hamartomatous polyposis and skin pigmentation (5). Hamartomatous polyps are typically large, but few in number; they may be present in both the small and large bowel. Skin pigmentation consists of freckles on the hands, perioral skin, periorbital skin, and buccal mucosa (5). Peutz-Jeghers syndrome is associated with an increased risk of colorectal, breast, pancreatic, gastric, ovarian, lung and small bowel malignancies (4).
Juvenile polyposis syndrome is a rare genetic condition inherited in an autosomal dominant manner. Patients with this syndrome develop hamartomatous polyps throughout their gastrointestinal tract, which usually form before age 20 (8). The number of polyps can vary from only a few to several hundred, even among members of the same family. Patients with juvenile polyposis syndrome are estimated to have a 10-50% risk of developing a gastrointestinal malignancy, with colorectal cancer being the most common type to occur (8).
Patients who have previously been diagnosed with colon adenomas or colorectal cancer are at increased risk of subsequently developing new colorectal cancer.
Chronic inflammatory bowel disease that involves the colon is associated with an increased risk of developing colorectal cancer. The extent to which risk is increased is determined by the extent of colon involved, the duration of the disease, the presence of dysplasia and other prognostic factors (9).
A sedentary lifestyle is associated with an increased risk of developing colorectal cancer, while physical activity is thought to be protective against the development of colorectal cancer (4).
Dietary factors are also associated with colorectal cancer. Frequent consumption of red meat is associated with an increased risk of colorectal cancer development, and an increased risk of dying from colorectal cancer (4). Consumption of processed meats (i.e. meats preserved by smoking ,curing, salting or the addition of preservatives such as nitrates/nitrites) are also associated with an increased risk of colorectal cancer, and the risk is thought to increase with greater amounts consumed (4). Diets low in fibre are also associated with an increased risk of colorectal cancer, while consumption of fruits and vegetables is thought to be protective (4).
Obesity is another lifestyle-associated risk factor for colorectal cancer. Individuals with a body mass index (BMI) in the obese range are thought to have a 1.5 times increased risk of developing colorectal cancer compared to those with a BMI in the normal range (4).
Alcohol use at a level greater than 3.5 drinks or more per day is associated with a 1.5 times increased risk of developing CRC compared to non-drinkers (4).
Prolonged cigarette smoking (> 35 pack years) has also been associated with a significantly increased risk of developing colorectal cancer (4,5).
New research in JAMA Oncology in November 2025, suggests that ultraprocessed foods may be a risk factor for colon cancer. The study looked at a cohort of 29 105 female participants, under the age of 50. They found that higher ultraprocessed food intake was associated with increased risk of early-onset colorectal conventional adenomas. Ultraprocessed foods were defined by the Nova classification (10).
For interest the article can be found here.
There is a higher risk of colorectal cancer in among members of certain ethnicities, including Ashkenazi Jews and African Americans, due to an increased incidence of genetic mutations that predispose to colorectal cancer (4).
Colorectal cancer also has a substantially higher rate of incidence and mortality in the developed Western world as compared to other geographic regions such as Asia and Africa. This is thought to be explained by the increased consumption of meats and sedentary lifestyle of economically privileged nations (5).
FIT is the preferred method of screening for average risk asymptomatic patients due to its convenience, safety, and cost-effectiveness (1,2).
FIT is preferred over guaiac-based fecal occult blood testing due to its greater specificity. FIT is positive only in the presence of human hemoglobin, while FOBT may be positive with any source of heme, including dietary and medication sources (1).
Colonoscopy is the gold-standard for adenoma detection and removal. It has excellent sensitivity and specificity, allows direct inspection of the entire colon, and enables biopsy and polypectomy at the time of diagnosis (1).
Disadvantages of colonoscopy include the invasive nature of the investigation, the requirement for sedation, and the risk of serious complications including bowel perforations (0.38 per 10,000). Colonoscopy is also a much more costly investigation (1).
Flexible sigmoidoscopy is an endoscopic procedure limited to the rectum and sigmoid colon. Advantages include the ability to perform biopsies and polypectomies, and the lack of need for sedation for most patients. Disadvantages include discomfort, risk of serious complications, and inability to examine the proximal colon (1).
CT colonography images the entire colon using CT imaging. It requires a thorough bowel preparation and insufflation with carbon dioxide gas through the rectum to allow visualization. Advantages include its minimally invasive nature, lack of need for sedation, and low complication rate compared to colonoscopy. Disadvantages include radiation exposure, high cost per procedure, reduced sensitivity for sessile (flat) polyps, and inability to perform biopsy or polypectomy (1).
Note screening does not apply to patients demonstrating new onset symptoms. Screening is for the general healthy population.
Screening is not recommended in this age group for average risk individuals.
Both the BC Cancer Agency and the British Columbia colorectal cancer screening guidelines (2024) recommend screening average risk asymptomatic patients aged 50-74 years with FIT every 2 years (1,3). Screening with FIT in this population has been associated with a reduction in mortality from colorectal cancer (1). Positive FIT should be followed up with colonoscopy for diagnostic investigation (4).
The guidelines also state that colonoscopy every 10 years is considered an acceptable alternative screening option for this population (1).
The Canadian Task Force on Preventive Health Care makes similar recommendations regarding FIT, but recommends flexible sigmoidoscopy rather than colonoscopy as an alternative screening method (2). They state that colonoscopy is not recommended as an alternative screening method due to increasing wait times, higher cost per procedure, and greater potential for harms with no proven advantage in terms of mortality reduction (2).
The BC Cancer Agency does not offer a screening recommendation for this age group, but states that there is limited evidence for screening outside the age range of 50-74 years (3).
The recommendation is that patients ages 75-85 years discuss the benefits and risks of screening with their primary care provider, and that a decision be made after consideration of the patient’s values, medical comorbidities, and life expectancy (1). The guidelines recommend that patients older than 85 years not be screened (1).
The Canadian Task Force on Preventive Health Care recommends not screening adults older than 75 years of age (2).
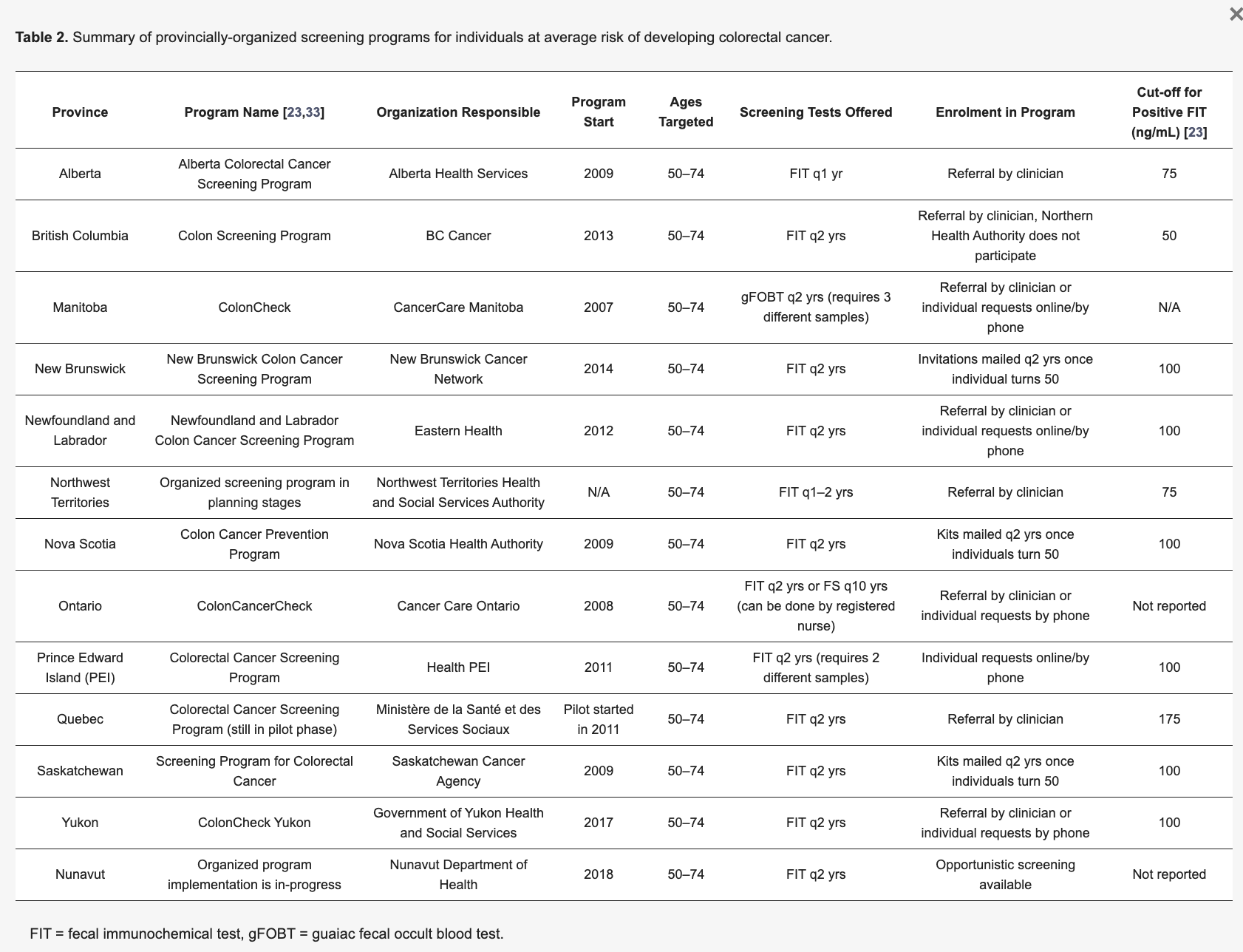
Screening guidelines vary by province. See the table below for more detailed information.
Patients are classified as increased risk if they have a personal history of adenoma or colorectal cancer, a significant family history of colorectal cancer, hereditary polyposis syndrome, or longstanding inflammatory bowel disease (1).
According to both the BC Cancer Agency and the BC guidelines for colorectal cancer screening, patients with a significant family history of colorectal cancer are recommended to undergo screening by colonoscopy every 5 years beginning at age 40 years, or 10 yrs earlier than the age of the youngest affected relative at diagnosis (1,3).
The BC Cancer Agency recommends that individuals with familial adenomatous polyposis (FAP) be screened by yearly flexible sigmoidoscopy beginning at puberty. Once polyposis is identified, prophylactic colectomy is recommended as the risk of developing colorectal cancer nears 100% (4).
All 1st degree relatives of individuals with a hereditary syndrome associated with colorectal cancer should be offered genetic counselling and testing. This should be carried out through a referral to the hereditary cancer programs in each province.
The BC guidelines also state that patients with inflammatory bowel disease with a duration of longer than eight years should undergo colonoscopy every 1-2 years with multiple biopsies taken to detect the presence of occult dysplasia (1). The BC Cancer Agency states that screening of individuals with IBD should be determined by the treating gastroenterologist due to variable risk associated with differing features of disease (4).
The BC Cancer Agency has developed an algorithm (July 2025) to determine next steps and future screening for individuals based on the number of pre-cancerous lesions and how high risk these lesions are (6).
This can be found here.
Hyperplastic polyps are benign polyps that have no potential for malignant transformation; they are not associated with an increased risk of colorectal cancer (1,2).
Inflammatory polyps are usually found in people with IBD involving the colon (1).
Hamartomas are also known as hamartomatous polyps. They are non-cancerous tumours made of overgrowth of normal cells. They are usually asymptomatic, and are not associated with malignant transformation or increased risk of colorectal cancer (1).
Lipomas are non-cancerous tumors that start in fat cells. They are usually asymptomatic but may require removal if they become large enough to cause bowel obstruction. They are not at risk for malignant transformation and are not associated with an increased risk of colorectal cancer (1).
Adenomas, or adenomatous polyps, are benign neoplasms that have potential for malignant transformation (2). This transformation usually occurs over many years, and is referred to as the ‘adenoma-carcinoma sequence’ (3). Adenomas are usually asymptomatic, but in some cases may cause rectal bleeding, fatigue, anemia or change in bowel habits.
All adenomas should be removed due to their risk of malignant transformation, and all patients with previous adenomas require ongoing colonoscopy for surveillance due to their elevated risk of developing new adenomas and colorectal cancer (4).
Adenomas are classified according to their morphologic features. They are usually polypoid, meaning that they stick out from the lining of the colon. Most adenomas are also pedunculated, meaning that they are mushroom-shaped with a stem/stalk. However, they may also be sessile, meaning that they lie flat against the colon wall and have no stalk. Adenomas may also be described as serrated, meaning that they have a saw-like border (1).
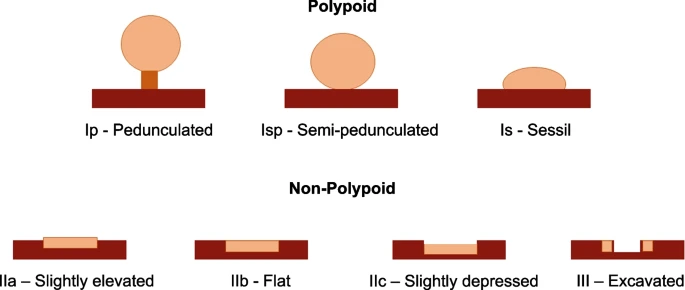
Adenomas are also classified according to their histopathological features. Tubular adenomas are the most common subtype, and are characterized by cells that look like tubes. Villous adenomas are characterized by their finger-like projections of cells. Tubulovillous adenomas have features of both tubular and villous growth patterns (1). Adenomas with villous growth patterns are at higher risk for malignant transformation (2).
Adenomas may also be classified as low, medium or high grade depending on the degree of dysplasia observed in histopathology. High-grade adenomas are associated with a greater risk of malignant transformation (2).
The risk of malignant transformation is greatest for adenomas with certain high-risk features (3):
The presence of multiple adenomas of any size is also associated with a greater risk of developing colorectal cancer (3).
Adenomas with any high-risk features should be followed up with repeat colonoscopy 6 months after polypectomy (4). Low-risk adenomas should be followed up with repeat colonoscopy in time-frame determined by the number of polyps (4).
Colorectal cancers are classified histopathologically by both type and grade.
Adenocarcinomas are by far the most common form of colorectal cancer, representing more than 95% of cases (1). These malignancies arise from gland cells that line the wall of the colon or rectum and produce mucus (1).
Adenocarcinomas may be unspecified (most common), or may be classified as one of two subtypes. Mucinous adenocarcinomas account for 10-15% of colorectal cancers, and are characterized by large amounts of extracellular mucin that comprises more than 50% of the tumour mass. They are associated with a poor response to chemotherapy and tend to present at a more advanced stage.
Signet-ring cell carcinomas account for 1-2% of all colorectal cancers, and are characterized by large amounts of intracellular mucin that comprises more than 50% of the tumour mass. The large quantity of intracellular mucin displaces the nucleus and cytoplasm to the sides of the cell, causing the cell to look like a signet ring on histopathological examination. Signet-ring cell carcinomas tend to be aggressive, and have a high potential for intramural spread and peritoneal carcinomatosis. Patients usually present at an advanced stage.
Rare forms of colorectal cancer other than adenocarcinoma may also occur, including squamous cell carcinoma, small cell carcinoma, medullary carcinoma, neuroendocrine tumours, soft tissue sarcoma and non-Hodgkin lymphoma (1).
Adenosquamous carcinoma represents 0.06% of all colorectal cancers (6). These are adenocarcinomas with areas of squamous differentiation. They often present at an advanced stage, behave aggressively, and are associated with a poor prognosis (6).
Small-cell carcinoma accounts for less than 1% of all colorectal cancers (7). These tumours are characterized by neuroendocrine features, and are associated with a poor prognosis (7).
Medullary carcinoma accounts for 3.6% of all colorectal cancers (8). This colorectal cancer subtype is associated with microsatellite instability and often occurs in patients with HNPCC. It has a more favourable prognosis than other colorectal cancer types (8).
Colorectal cancers may also be classified on the basis of histopathological grade, which is determined by the degree to which the cells appear abnormal on pathology.
For colorectal cancers, grading is based primarily on the presence or absence of well-formed glands. Low-grade tumours are those that contain well-formed glandular tissue, while high-grade tumours do not have organized glandular structures.
Grading:
The terms “low-grade” and “high-grade” refer to the grades G1-G2 and G3-G4 respectively.
Colorectal cancer is often asymptomatic, which is why screening is critical to detect pre-malignant and malignant lesions as early as possible (1).
Colorectal cancer may present with a number of symptoms, signs, or complications, including (1):
Patients who are symptomatic due to obstruction or perforation at the time of diagnosis carry a worse prognosis than patients who are asymptomatic.
20% of patients with colorectal cancer have metastatic disease at the time of presentation. The most common sites of metastatic spread in colorectal cancer are the regional lymph nodes, liver, lungs and peritoneum; the liver is often the first distant site involved. Colorectal cancer may therefore also present with signs and symptoms concerning for metastatic disease, including:
A more detailed description of the signs and symptoms associated with colon cancer, including their pathogenesis, is included in Table 1 below.

Physical examination should include a complete abdominal exam, including palpation for masses and hepatomegaly. The patient should also be examined for lymphadenopathy (1).
A digital rectal exam should be performed. If a lesion is palpable on a digital rectal exam, the size of the tumour, mobility, and distance from the anal verge should be documented (2).
For female patients, a pelvic and rectovaginal examination should be performed to look for local invasion of the tumour into pelvic structures (e.g., vagina, rectovaginal septum) (2).
There are no blood tests that are effective at detecting colorectal cancer. However, laboratory investigations may be performed to assess for complications of colorectal cancer and to assist with clinical staging of the malignancy.
A CBC should be performed to assess for the presence of a microcytic anemia due to chronic blood losses (1). FOBT or FIT may also be performed if these were not done as a component of screening.
AST, ALT should be obtained as an indicator of metastatic spread to the liver. Alkaline phosphatase may be obtained as an indicator of metastatic spread to bone. Bilirubin is critical to assess liver function and determine a patient's eligibility for treatment.
A carcinoembryonic antigen (CEA) level should also be obtained preoperatively to provide a baseline level (1,3). CEA is a tumour marker that is often used as a component of surveillance following surgery for colorectal cancer. The level should drop to near zero after surgery, and any increase in level thereafter could be indicative of a recurrence of the cancer (1).
Electrolytes, creatinine and urea may also be obtained in order to assess renal function prior to proceeding with any surgery or chemotherapy.
Individuals with an abnormal screening result or symptoms suspicious for colorectal cancer should undergo colonoscopy as the diagnostic test of choice. Colonoscopy allows direct visualization of the entire colon, as well as procedures such as biopsy and polypectomy.
However, approximately 5% of patients will have colon cancers that cannot be viewed using colonoscopy, due to an obstruction, tortuous colon, or poor bowel prep. In these patients, alternate choices for diagnostic imaging include CT colonography and double contrast barium enema. In double contrast barium enema, the patient undergoes an air enema followed by a barium enema. Multiple X-ray images are then taken to visualize any lesions present in the lumen of the colon.
A preoperative CT scan of the chest, abdomen and pelvis is recommended to exclude the presence of distant metastases and to provide a baseline for surveillance following treatment (3). PET scans are not usually recommended for the purposes of initial staging, but may be used in certain cases (3).
Ultrasound may be used to detect metastatic spread to the liver.
MRI of the pelvis is important to assess the depth of invasion of the cancer, its relationship to important landmarks such as the mesorectal fascia, and involvement of pelvic lymph nodes.
Transrectal endoscopic ultrasound may also be used for rectal cancers, and may have better resolution than MRI in very early stage cancers. Endoscopic ultrasound allows ultrasound-guided biopsies to be taken, and also provides important information for use in staging of the malignancy (3). However, the sensitivity of this investigation for lymphadenopathy is low, and thus a negative ultrasound study does not rule out lymph node involvement.
A chest x-ray may be performed to check if colorectal cancer has spread to the lungs in patients complaining of shortness of breath or other suspicious symptoms (3). This may not be necessary if a CT-Chest is being performed as a component of staging.
Biopsy is essential for confirming the diagnosis of colorectal cancer (1). Biopsies are usually obtained during colonoscopy (1). The pathologist will also determine the histopathological type and grade of the cancer (see Classification section for details).
Molecular testing may also be performed on the tumor sample. Testing for KRAS, NRAS, and BRAF mutations is often performed, as the presence of these mutations suggests that the tumor is unlikely to respond to targeted therapy drugs such as cetuximab and panitumumab (1).
The most widely accepted system for colorectal cancer staging is TNM staging, as outlined by the American Joint Committee on Cancer (AJCC) (1). This system stages colorectal cancer according to three primary features. The first feature is the tumor size, represented by ‘T’. The second feature is the extent of spread to regional lymph nodes, represented by ‘N’, which can be determined either clinically (‘cN’) or pathologically (‘pN’). The third feature used to classify colorectal cancer is the presence of any distal metastasis, represented by ‘M’. On the basis of these features, colorectal cancer is assigned a TNM status, which correlates to a certain stage of colorectal cancer.
For information on staging please refer to http://www.bccancer.bc.ca/books/Documents/Gastrointestinal/Stagingcolon1.pdf
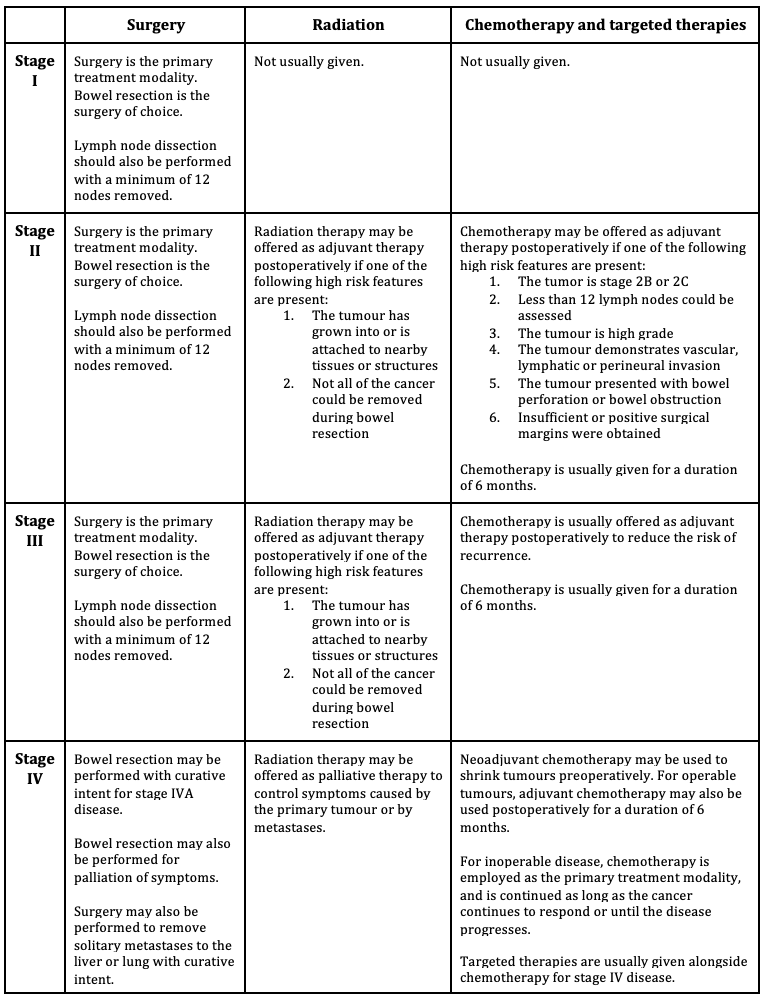
Colorectal cancers are managed through the combined use of several treatment modalities, including surgery, chemotherapy, radiation and targeted therapies (1).
Surgery is the primary treatment modality employed to manage colorectal cancers that are amenable to curative therapy (1). Several different surgical techniques are employed in the management of colorectal cancers depending on the clinical situation; these are outlined below.
In general, the surgical standard of care for patients with localized disease is resection of the involved segment of the colon, along with a margin of healthy tissue and a sample of at least twelve lymph nodes (2). Factors considered in determining the surgical approach include the distance of the distal edge of the tumour from the anal verge, body habitus, tumour size, and patient comorbidity.
For patients with metastatic disease, resection of an asymptomatic primary tumour is not routinely recommended, but surgical procedures may be performed for other indications (2).
Local excision refers to the removal of a polyp or tumour and a margin of healthy tissue (1). It is primarily used to remove premalignant adenomatous polyps and low-risk stage 1 colorectal cancers (1). When performed for this indication, local excision is also known as a polypectomy, and is usually performed during colonoscopy. To be a candidate for local excision, patients must have colorectal cancer with low-risk features, including a well-differentiated appearance on histology, size < 3 cm, and no lymphatic or vascular invasion.
Local excision may also be performed to remove stage 0 or stage 1 tumours in the distal rectum using special tools placed in the anus. This procedure is known as a local transanal excision (1).
Bowel resection is the most common type of surgery performed for colorectal cancer. In this surgery, a segment of the bowel is removed, along with a margin of healthy tissue and a minimum of twelve lymph nodes (1). The surgeon should aim to remove a margin of healthy tissue of 5 cm both proximal and distal to the lesion (2).
The various types of bowel resection are named for the segment of bowel removed, and include right hemicolectomy, transverse colectomy, left hemicolectomy, sigmoid colectomy, low anterior resection (removes the most distal segment of the colon and part of the rectum), and abdominoperineal resection (removes the rectum, anal canal, and surrounding muscles) (1).
Laparoscopic resection can be performed with equivalent efficacy and decreased recovery time. However, it requires an experienced surgeon capable of performing a sufficiently thorough abdominal examination (2).
Bowel resection is associated with a number of possible side effects, including (1):
In particular, abdominoperineal resection is associated with a high rate of sexual and urinary side effects.
Pelvic exenteration is a procedure sometimes used to treat stage IV or recurrent rectal cancer that has spread to nearby organs. It involves removal of the rectum, lymph nodes, reproductive organs, and sometimes the bladder (1).
Colostomy is a procedure in which a stoma (opening) is created to connect the lumen of the colon to the outside of the body. A temporary colostomy may be required if primary anastomosis is not possible following a bowel resection. In this case, the colostomy is removed once secondary anastomosis of the remaining bowel segments is performed (1).
Permanent colostomies are not commonly required following bowel resections; however, they are required after abdominoperineal resection (1).
Lymph node dissection is performed via the en bloc removal of a section of mesentery containing at least twelve lymph nodes. Removal of fewer lymph nodes is inadequate, and can result in increased mortality due to inaccurate staging.
Colorectal cancers with solitary metastatic lesions to the liver and lungs are potentially curable by surgical excision (1). There are reports of 25-30% disease free survival rates after excision of isolated liver metastases (3).
Surgical procedures employed in the palliative management of colorectal cancers include bowel resection, and bypass with diverting colostomy. Indications for these procedures include colorectal cancer causing significant local symptoms such as pain or bowel obstruction.
Chemotherapy is often given after surgery as adjuvant therapy for stage II and III colon cancer. It is also the primary treatment modality used for inoperable stage IV or recurrent colon cancers (1). Chemotherapy may also be given in combination with radiation therapy for stage II and III rectal cancer, either before or after surgery; this is referred to as chemoradiation.
Chemotherapy regimens for colorectal cancer are associated with many potential side effects, including (1):
External beam radiation therapy is the most common radiation modality employed, but brachytherapy may be used in certain clinical situations (1).
Radiation may be given as neoadjuvant therapy prior to surgery to reduce tumour size and improve operability (1). It may also be given after surgery as adjuvant therapy to reduce the risk of locoregional recurrence (1). Radiation therapy is also sometimes used as a palliative therapy to treat symptomatic metastatic disease in the abdomen, pelvis, bone or brain (1).
Radiation therapy for colorectal cancer may be associated with side effects, including (1):
Targeted therapies are medications used to treat colorectal cancer, and an area of growing research. They include monoclonal antibodies that target antigens expressed by colorectal cancers and small molecule tyrosine inhibitors that target specific signalling pathways. They may have fewer side effects than traditional chemotherapy. They may be given in concert with chemotherapy. Agents used in the treatment of colorectal cancer include cetuximab, panitumumab, bevacizumab, and fruquintinib, among others (1).
Side effects of targeted therapies for colorectal cancer include (1):
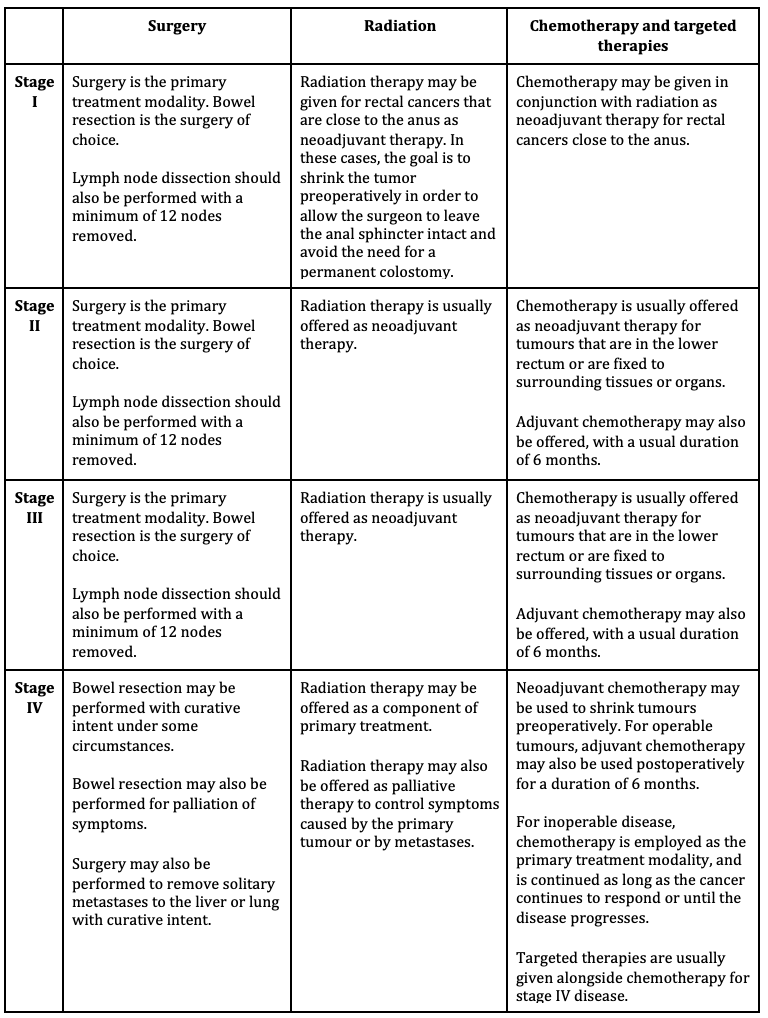
The approach to management of early stage colorectal cancer is different depending on whether the tumour is located in the colon, or in the rectum. For the purpose of managing colorectal cancer, the division between the colon and rectum is located at the point of the peritoneal reflection.
One of the major differences between the management of colon cancer and rectal cancer is that adjuvant therapy for colon cancer consists mainly of chemotherapy due to increased likelihood of distant metastasis, while for rectal cancer both radiation and chemotherapy are used due to local and distant metastasis .
As with all other types of cancer, application of general cancer management principles involves consideration of patient, tumour and treatment factors when deciding the best course of treatment for each individual case.
Please see the updated American Cancer Guidelines 2022 for more details.
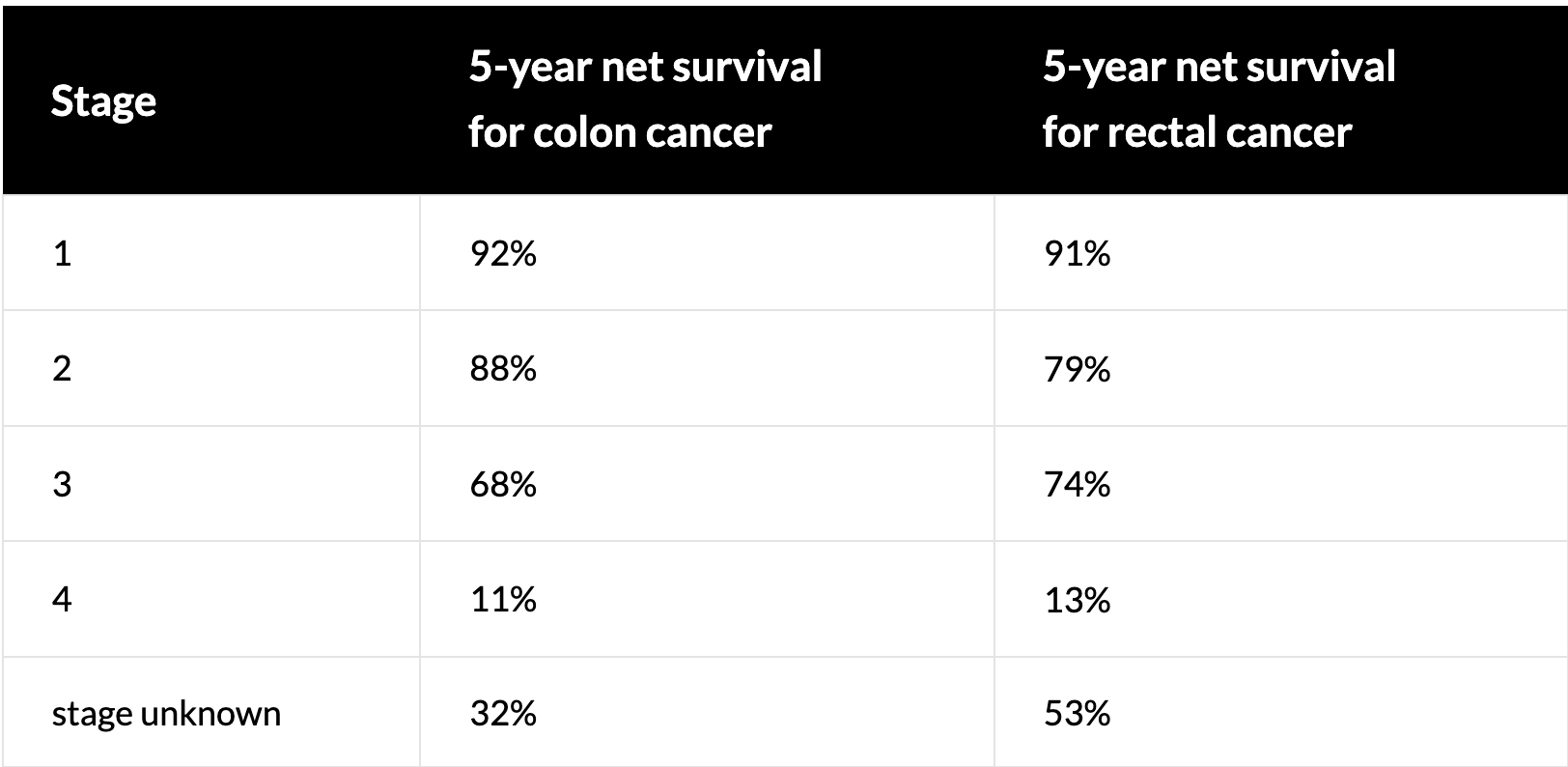
The overall 5-year survival for colorectal cancer in Canada is 67% (4). However, the prognosis of individual patients with colorectal cancer is influenced significantly by various prognostic factors.
The most important prognostic factor for colorectal cancer is the stage of disease, with more advanced disease having poorer outcomes (2). Localized colorectal cancer has a 90% 5-year survival rate, while regional and distant metastatic disease are associated with 71% and 13% 5-year survival rates respectively (2).
Pathohistological factors associated with a worse prognosis include positive nodal status, greater depth of invasion, positive surgical margins, lymphovascular invasion, higher pathological grade, and certain types of tumour (mucinous adenocarcinoma, signet ring cell adenocarcinoma, and small cell carcinoma) (2). Certain genetic changes have also been associated with a poor prognosis, including mutations in the KRAS, NRAS, DCC and BRAF genes (2).
Patients with higher preoperative CEA levels, or who present with bowel obstruction or perforation, have also been found to have worse prognosis (2).
Patient Resources: https://www.colorectalcancercanada.com/colorectal-cancer/treatments/
Follow-up is typically performed by both the treating specialists and the patient’s family physician. The most important phase of follow-up is the first three years after primary treatment, as this is the time in which the majority of recurrences will occur (1).
After primary treatment for colorectal cancer is completed, patients should be seen in follow-up every 3-6 months for a duration of two years, then every 6 months for a total duration of 5 years (1,2). After this period, patients should continue to be seen in follow-up annually (3).
At each follow-up visit, the following tasks should be performed:
History: Ask about gastrointestinal and constitutional symptoms, as well as nutritional status (3).
Physical Exmination: Complete abdominal exam, palpation for lymphadenopathy, palpation for hepatomegaly, auscultation of the lungs, and digital rectal exam (2,3). Particular attention should be paid to the left supraclavicular fossa, which is the location of Virchow’s node, a common location for metastases from gastrointestinal cancers.
Labs: Patients with a history of stage II or III colorectal cancer should have a serum CEA level obtained prior to every visit as a component of surveillance (1). If CEA is elevated, the level should be repeated within 28 days (3). If persistently elevated, imaging of the chest and abdomen should be obtained to assess for recurrence.
Colonoscopy: Repeat colonoscopy should be performed within 12 months of completing primary treatment. If normal, colonoscopy should then be repeated in three years time and then every five years thereafter (1,3).
Sigmoidoscopy: Patients with rectal cancer should undergo flexible sigmoidoscopy every 6 months for 2-5 years, particularly if they did not receive radiation therapy (2). The goal is to assess recurrent disease at the site of anastomosis.
Liver Imaging: Imaging should be performed by either US or CT every 6 months for the first three years, and then annually for two additional years (1). However, this does vary by province so confirm with your local health authority. This component of surveillance is only indicated in those patients who would be candidates for resection of isolated liver metastases.
CT Chest and Abdomen: For stage I or II colorectal cancers, CT chest and abdomen should be obtained twice within the first three years, with 12 months and 36 months being the suggested times (2).For stage III or IV colorectal cancers, follow-up imaging with CT chest and abdomen should be performed annually for 3-5 years (2).
Chest X-Ray: For patients with rectal cancers, chest x-ray should be performed annually for three years to assess for metastatic spread to the lungs (2).
The increasing rate of colorectal cancer in those under 50 years old in Canada is concerning, as this age group is not included in the current screening guidelines (1). This rise primarily affects individuals aged 40-49 (1). No changes have yet been made to Canadian screening guidelines. However, U.S. screening guidelines have been modified to start at 45 years of age to include this cohort.
The cause for this demographic shift has not been elucidated but plausible explanations include a Western-style diet, higher rates of obesity especially in adolescents, physical inactivity, and antibiotic use from the prenatal to adolescent period (2) . Primary prevention is focused on modifying risk factors such as diet, exercise, smoking cessation, and limiting alcohol consumption; however, further research is needed to determine risk factors for early-onset colorectal cancer (2).
Use your mouse to click through the slides and answer each question in the text box provided.
Note: This case can be completed on an iPad. To do this download the (free) Articulate Mobile Player for the iPad by clicking here.
